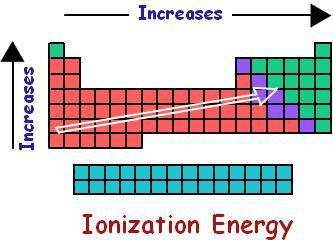
The first ionization energy of an element is the energy required to remove an electron from a gaseous atom of an element to produce a +1 ion:
M(g) + energy ---> M ^(+) (g) + e -
How do you think the activity of an element ought to be related to its first ionization energy? Predict a decreasing order of reactivity of the above elements based on their first ionization energies.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:40
In which environment would primary succession occur? a forest with a few remaining trees after a recent wildfire an area of exposed rock after a glacier melts away beach that is exposed to the air at low tide an abandoned baseball field in a small town
Answers: 1

Chemistry, 22.06.2019 07:30
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 11:00
The twister and runaway train are two coasters at the same amusement park. both coasters start at the same height. the coaster for the twister is twice the mass of the coaster for the runaway train. which roller coaster has greater gravitational potential energy at the start of the ride?
Answers: 1

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3
You know the right answer?
The first ionization energy of an element is the energy required to remove an electron from a gaseou...
Questions


Mathematics, 03.09.2020 14:01



Computers and Technology, 03.09.2020 14:01


English, 03.09.2020 14:01

Health, 03.09.2020 14:01


Mathematics, 03.09.2020 14:01

History, 03.09.2020 14:01

English, 03.09.2020 14:01


Physics, 03.09.2020 14:01


Mathematics, 03.09.2020 14:01



Computers and Technology, 03.09.2020 14:01

Mathematics, 03.09.2020 14:01