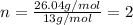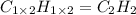When 2.935 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 9.920 grams of CO2 and 2.031 grams of H2O were produced. In a separate experiment, the molar mass of the compound was found to be 26.04 g/mol. Determine the empirical formula and the molecular formula of the hydrocarbon.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 06:30
Three cards with holes are arranged in a straight line. a light is shined through the first card’s hole and travels through all three cards. what does this tell you about light rays? a) that light is reflected b) that light is refractive c) that light travels in a straight line d) that light does not travel in a straight line
Answers: 1

Chemistry, 22.06.2019 10:40
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1
You know the right answer?
When 2.935 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 9.920 grams...
Questions


History, 24.11.2020 23:50



Computers and Technology, 25.11.2020 01:00

Arts, 25.11.2020 01:00

Mathematics, 25.11.2020 01:00

Health, 25.11.2020 01:00

History, 25.11.2020 01:00

History, 25.11.2020 01:00

Mathematics, 25.11.2020 01:00


English, 25.11.2020 01:00


Mathematics, 25.11.2020 01:00

Mathematics, 25.11.2020 01:00


Mathematics, 25.11.2020 01:00

English, 25.11.2020 01:00

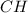 and
and 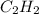 respectively.
respectively.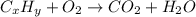
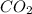 = 9.920 g
= 9.920 g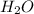 = 2.031 g
= 2.031 g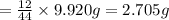 of carbon will be contained.
of carbon will be contained.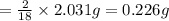 of hydrogen will be contained.
of hydrogen will be contained.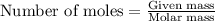 ......(1)
......(1)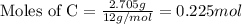
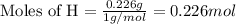
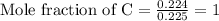
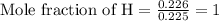
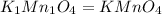
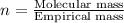 .....(2)
.....(2)