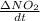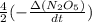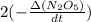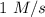Chemistry, 10.06.2021 03:50 tatibean26
If the initial concentration of N2O5 was 0.500 M and the concentration of N2O5 was 0.450 M after 0.100 s, what is the average rate of NO2 formation during the first 100 milliseconds

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
If the initial concentration of N2O5 was 0.500 M and the concentration of N2O5 was 0.450 M after 0.1...
Questions

History, 10.01.2020 23:31

Chemistry, 10.01.2020 23:31




Mathematics, 10.01.2020 23:31




Mathematics, 10.01.2020 23:31

Mathematics, 10.01.2020 23:31


Mathematics, 10.01.2020 23:31

Mathematics, 10.01.2020 23:31

Social Studies, 10.01.2020 23:31


Mathematics, 10.01.2020 23:31

Mathematics, 10.01.2020 23:31


Mathematics, 10.01.2020 23:31

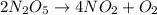

 formation:
formation: