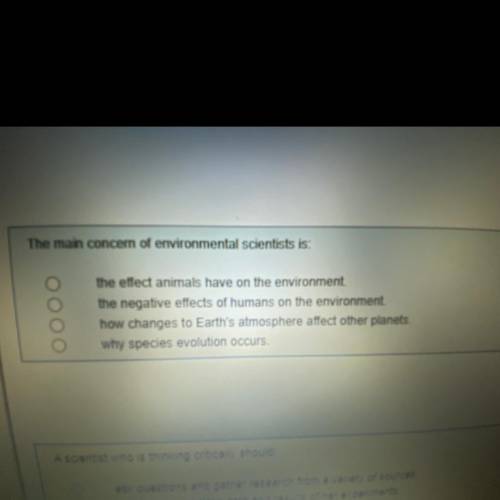
Plz help with quiz I’ll mark brainliest
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1


Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
You know the right answer?
Questions

Mathematics, 20.11.2020 17:40

Mathematics, 20.11.2020 17:40



Mathematics, 20.11.2020 17:40

Mathematics, 20.11.2020 17:40


Biology, 20.11.2020 17:40


Mathematics, 20.11.2020 17:40

Mathematics, 20.11.2020 17:40

Chemistry, 20.11.2020 17:40


Advanced Placement (AP), 20.11.2020 17:40


Biology, 20.11.2020 17:40

English, 20.11.2020 17:40


Mathematics, 20.11.2020 17:40

Mathematics, 20.11.2020 17:40