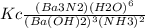The expression for the equilibrium constant for the reaction:
Ba3N2 (aq) + 6 H2O(1)-
3 Ba(OH)...

Chemistry, 07.06.2021 22:50 teddybear196510
The expression for the equilibrium constant for the reaction:
Ba3N2 (aq) + 6 H2O(1)-
3 Ba(OH)2 (aq) + 2 NH3 (9)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2


Chemistry, 23.06.2019 01:00
You wish to prepare a buffer consisting of acetic acid and sodium acetate with a total acetic acetate plus acetate concentration of 250 mm and a ph of 5. what concentrations of acetic acid and sodium acetate should you use
Answers: 1

Chemistry, 23.06.2019 04:00
Which method would be best to separate a mixture of sand and gravel
Answers: 1
You know the right answer?
Questions

Mathematics, 19.12.2019 13:31

Social Studies, 19.12.2019 13:31

World Languages, 19.12.2019 13:31







Chemistry, 19.12.2019 13:31

Social Studies, 19.12.2019 13:31




History, 19.12.2019 13:31

Social Studies, 19.12.2019 13:31



History, 19.12.2019 13:31

Health, 19.12.2019 13:31