For the equilibrium that exists in an aqueous solution of nitrous acid (HNO2, a weak acid), the equilibrium constant expression is:
a. K = [ H+] [NO2-] / [HNO2]
b. K = [ H+] [N] [O]2 / [HNO2]
c. K = [ H+] [NO2-] / [HNO2]
d. K = [H+]2 [NO2-] / [HNO2]
e. None of these

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 22.06.2019 22:30
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3
You know the right answer?
For the equilibrium that exists in an aqueous solution of nitrous acid (HNO2, a weak acid), the equi...
Questions

Mathematics, 24.03.2021 03:30

Mathematics, 24.03.2021 03:30


Mathematics, 24.03.2021 03:30

Arts, 24.03.2021 03:30

Mathematics, 24.03.2021 03:30

Business, 24.03.2021 03:30

Mathematics, 24.03.2021 03:30

Physics, 24.03.2021 03:30

Arts, 24.03.2021 03:30



Mathematics, 24.03.2021 03:30

Mathematics, 24.03.2021 03:30



Chemistry, 24.03.2021 03:30


Biology, 24.03.2021 03:30

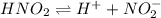
![K = \frac{[H^{+}][NO^{-}_{2}]}{[HNO_{2}]}](/tpl/images/1365/7220/b80db.png)