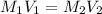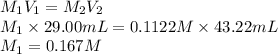Chemistry, 07.06.2021 15:30 biggiecheese93
A 29.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1122 M KOH solution. A volume of 43.22 mL of KOH was required to reach the equivalence point. What is the concentration of the unknown H2SO4 solution? Express your answer using four significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
If a 12-v battery is connected to a circuit that has a current of 3.0 a, what is the total resistance in the circuit? 36 ohms 4 ohms 0.25 ohms
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2

Chemistry, 22.06.2019 20:00
Which of the following would not diffuse through the plasma membrane by means of simple diffusion? 1 oxygen 2 glucose 3 a steroid hormone 4 a lipid soluble vitamin
Answers: 3

Chemistry, 23.06.2019 00:00
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2
You know the right answer?
A 29.00 mL sample of an H2SO4 solution of unknown concentration is titrated with a 0.1122 M KOH solu...
Questions



Mathematics, 17.10.2020 18:01


Mathematics, 17.10.2020 18:01

Mathematics, 17.10.2020 18:01


Mathematics, 17.10.2020 18:01






Mathematics, 17.10.2020 18:01



Health, 17.10.2020 18:01

Mathematics, 17.10.2020 18:01

Mathematics, 17.10.2020 18:01

Mathematics, 17.10.2020 18:01

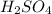 solution is 0.167 M.
solution is 0.167 M.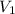 = 29.00 mL,
= 29.00 mL, 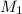 = ?
= ?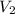 = 43.22 mL,
= 43.22 mL, 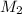 = 0.1122 M
= 0.1122 M