Aluminium is extracted from aluminium oxide using electrolysis.
Calculate the mass, in grams, of aluminium formed when a charge of 20 faradays
is passed through aluminium oxide dissolved in molten cryolite.
The ionic half-equation for the formation of aluminium is
Al3+ + 3e + Al

Answers: 1


Another question on Chemistry

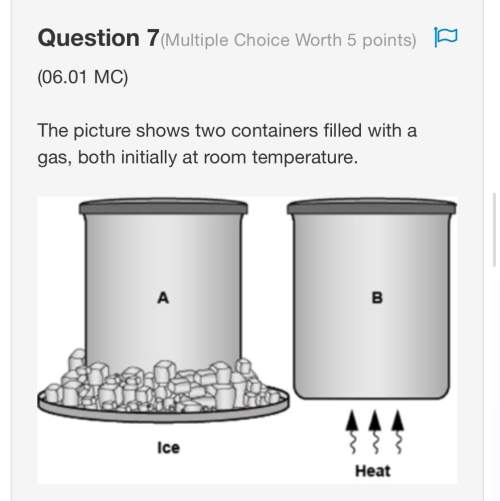
Chemistry, 22.06.2019 00:00
Acurium-245 nucleus is hit with a neutron and changes as shown by the equation. complete the equation by filling in the missing parts. 52
Answers: 2

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1
You know the right answer?
Aluminium is extracted from aluminium oxide using electrolysis.
Calculate the mass, in grams, of al...
Questions




Computers and Technology, 28.08.2019 17:30

History, 28.08.2019 17:30

Mathematics, 28.08.2019 17:30


Arts, 28.08.2019 17:30







Mathematics, 28.08.2019 17:30

English, 28.08.2019 17:30