
Chemistry, 05.06.2021 22:50 Kaysofine11icloudcom
What is the limiting reactant in a reaction where 10.0 mol of iron is treated with 12.0 mol of bromine? The product that forms is FeBr3. First, write and balance the chemical equation. Next, calculate the moles of FeBr3 that can be made from 10.0 mol of Fe. Then calculate the moles of FeBr3 that can be made from 12.0 mol of bromine. The smaller amount of FeBr3 reveals the limiting reactant.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
This chart represents the melting point of several substance. what besy explains the high melting point of the salt?
Answers: 2

Chemistry, 22.06.2019 08:40
What is the value of keq for the reaction expressed in scientific notation?
Answers: 1


Chemistry, 22.06.2019 11:00
An object becomes electrically charged when: electrons are created in it electrons from it are destroyed electrons are transferred to it protons from it are destroyed protons are created in it
Answers: 1
You know the right answer?
What is the limiting reactant in a reaction where 10.0 mol of iron is treated with 12.0 mol of bromi...
Questions


Biology, 17.06.2021 20:30

Mathematics, 17.06.2021 20:30

Computers and Technology, 17.06.2021 20:30


Mathematics, 17.06.2021 20:30





Advanced Placement (AP), 17.06.2021 20:30



Mathematics, 17.06.2021 20:30




Chemistry, 17.06.2021 20:30

History, 17.06.2021 20:30

Mathematics, 17.06.2021 20:30

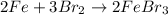
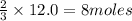 of iron
of iron

