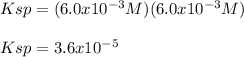Chemistry, 05.06.2021 03:40 hogwartsalicia
Solid strontium chromate, SrCrO4, dissolves into its respective ions at 25°C. Suppose that in a particular solution, [Sr^2+]=6.0x10^-3M. Find the value of Ksp.
A) 3.6 x 10^-5
B) 6.0 x 10^-3
C) 3.6 x 10^-2
D) 7.7 x 10^-2

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 15:30
Count the number of each type of atom in the equation below, and then balance the equation. write in the numbers of atoms and coefficients. add a 1 if there should be no coefficient. cs2(l) + o2(g) → co2(g) + so2(g) c [ ] s [ ] o > c [ ] s [ ] o [ ] cs2(l) + [ ] o2(g) > [ ] co2(g) + [ ] so2(g)
Answers: 3

Chemistry, 23.06.2019 02:30
Apound is approximately 0.45 kilogram. a persons weighs 87 kilograms. what is the persons’s weight, in pounds, when expressed to the correct number of significant figures
Answers: 1

Chemistry, 23.06.2019 03:20
What kind of intermolecular forces act between a hydrogen fluoride molecule and a hydrogen peroxide molecule? note: if there is more than one type of intermolecular force that acts, be sure to list them all, with a comma between the name of each force.
Answers: 1
You know the right answer?
Solid strontium chromate, SrCrO4, dissolves into its respective ions at 25°C. Suppose that in a part...
Questions


















Business, 06.07.2019 13:20

Chemistry, 06.07.2019 13:20

![Ksp=[Sr^{2+}][CrO_4^{2-}]](/tpl/images/1363/6886/12f99.png)