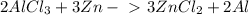Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:40
Pastoral farming is best described as a. a method of raising livestock and moving herds b. an african method of agriculture c. a method of cultivating crops on poor soils d. a common method of desert farming select the best answer from the choices provided a b c d
Answers: 2

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2

Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
You know the right answer?
How many moles of ZnCl2(little 2) are in 156 grams of ZnCl2 (little 2)...
Questions

History, 02.09.2019 17:50

History, 02.09.2019 17:50

Mathematics, 02.09.2019 17:50

Chemistry, 02.09.2019 17:50



Biology, 02.09.2019 17:50



Computers and Technology, 02.09.2019 17:50


Computers and Technology, 02.09.2019 17:50


Chemistry, 02.09.2019 17:50


Social Studies, 02.09.2019 17:50

Mathematics, 02.09.2019 17:50

English, 02.09.2019 17:50

Biology, 02.09.2019 17:50