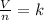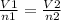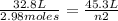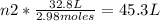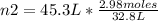Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:50
An engineering team designs a new rocket that is faster and lighter than any other model being produced. however, the materials end up being so expensive that no company can afford to buy them. which step of the engineering process should have addressed this problem? a. know the background. b. evaluate the results. c. identify a need. d. do the work.
Answers: 2

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3

Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
You know the right answer?
2.98 moles of H2 at 35°C and 2.3 atm are in a 32.8 L container. How many moles of H2 are in a 45.3 L...
Questions



Business, 19.07.2019 18:00

English, 19.07.2019 18:00

Biology, 19.07.2019 18:00

English, 19.07.2019 18:00

English, 19.07.2019 18:00

Mathematics, 19.07.2019 18:00

Mathematics, 19.07.2019 18:00

English, 19.07.2019 18:00

Business, 19.07.2019 18:00

History, 19.07.2019 18:00

English, 19.07.2019 18:00

Computers and Technology, 19.07.2019 18:00




Advanced Placement (AP), 19.07.2019 18:00

Spanish, 19.07.2019 18:00

Mathematics, 19.07.2019 18:00