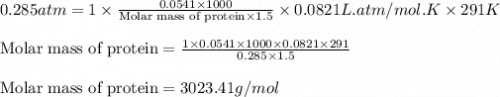Northern cod produce proteins that protect their cells from damage caused by subzero temperatures. Measurements of the osmotic pressure for two "antifreeze" proteins at 18°C yielded the data listed below. Use this information to calculate the molar mass for each of the proteins. Assume these proteins are nonelectrolytes and use the value i = 1.
Required:
If a 54.1 mg sample of protein A in 1.5 mL of water has an osmotic pressure of 0.285 atm, what is the molar mass of protein A?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:30
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3


Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 22.06.2019 17:30
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2
You know the right answer?
Northern cod produce proteins that protect their cells from damage caused by subzero temperatures. M...
Questions

Social Studies, 19.09.2019 02:00

Mathematics, 19.09.2019 02:00

Physics, 19.09.2019 02:00

Mathematics, 19.09.2019 02:00


Biology, 19.09.2019 02:00

Spanish, 19.09.2019 02:00


Mathematics, 19.09.2019 02:00


Mathematics, 19.09.2019 02:00


Social Studies, 19.09.2019 02:00



Chemistry, 19.09.2019 02:00




Biology, 19.09.2019 02:00

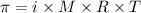
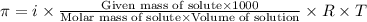 .....(1)
.....(1)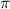 = osmotic pressure = 0.285 atm
= osmotic pressure = 0.285 atm![18^oC=[18+273]=291K](/tpl/images/1361/3277/7621e.png)