Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
It takes 945.kj/mol to break a nitrogen-nitrogen triple bond. calculate the maximum wavelength of light for which a nitrogen-nitrogen triple bond could be broken by absorbing a single photon.
Answers: 3

Chemistry, 22.06.2019 00:40
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1
You know the right answer?
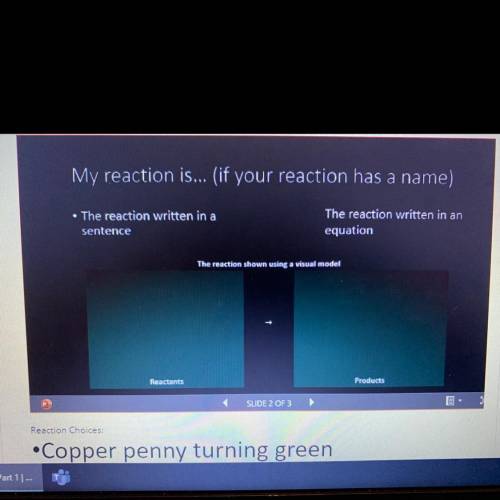
Please answer these questions and show me how the model too! This is for COPPER PENNY TURNING GREEN...
Questions

Chemistry, 06.11.2019 08:31

History, 06.11.2019 08:31


Business, 06.11.2019 08:31

Mathematics, 06.11.2019 08:31


Mathematics, 06.11.2019 08:31

English, 06.11.2019 08:31


Social Studies, 06.11.2019 08:31


Mathematics, 06.11.2019 08:31


Mathematics, 06.11.2019 08:31

History, 06.11.2019 08:31

History, 06.11.2019 08:31

Social Studies, 06.11.2019 08:31