
Chemistry, 27.05.2021 16:10 victoria1831
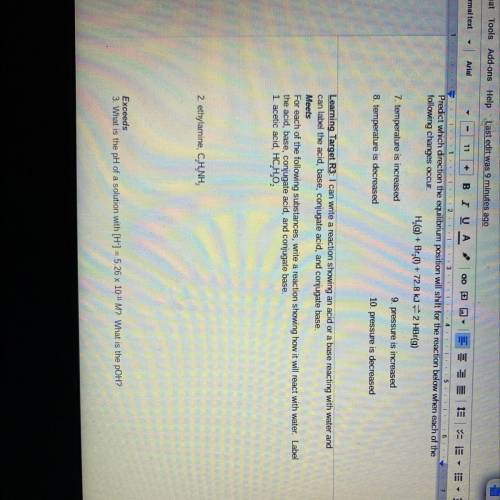
For each of the following substances, write a reaction showing how it will react with water. Label
the acid, base, conjugate acid, and conjugate base.
1. acetic acid, HC, H,02
2. ethylamine, C. H.NH,

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
12. complete each of the following word equations for synthesis reactions. a. sodium + oxygen -> b. magnesium + fluorine -> 13. complete and balance the equations for the decomposition reactions. a. hgo -> [with the triangle heat symbol above the arrow] b. h2o(l) -> [with "electricity" written above the arrow]
Answers: 1

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3
You know the right answer?
For each of the following substances, write a reaction showing how it will react with water. Label...
Questions


Mathematics, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00



Chemistry, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00


History, 21.11.2020 14:00





Physics, 21.11.2020 14:00

English, 21.11.2020 14:00

Geography, 21.11.2020 14:00

Mathematics, 21.11.2020 14:00

English, 21.11.2020 14:00



