Chemistry, 25.05.2021 21:20 martinbricein10
Given that ammonia (NH3) acts as a weak base in water with a Kb of 1.8 x 10-5 at 298.0 K, calculate the pH at 298.0 K of a solution prepared by mixing 100.0 mL of a 0.050 M aqueous solution of ammonia with 20.0 mL of a 1.00 M aqueous solution of nitric acid. The final volume of the solution is 120.0 mL.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:00
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1

Chemistry, 22.06.2019 23:40
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2
You know the right answer?
Given that ammonia (NH3) acts as a weak base in water with a Kb of 1.8 x 10-5 at 298.0 K, calculate...
Questions

Mathematics, 29.05.2020 11:57

Business, 29.05.2020 11:57


Mathematics, 29.05.2020 11:57


Mathematics, 29.05.2020 11:58














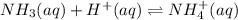
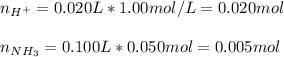
![[H^+]=\frac{0.015mol}{0.120L}=0.125M](/tpl/images/1347/3045/b1ea1.png)