Two water solutions that have a density of
1.00 g/mL are mixed, and a reaction occurs.
The te...

Chemistry, 25.05.2021 08:50 llnapier8924
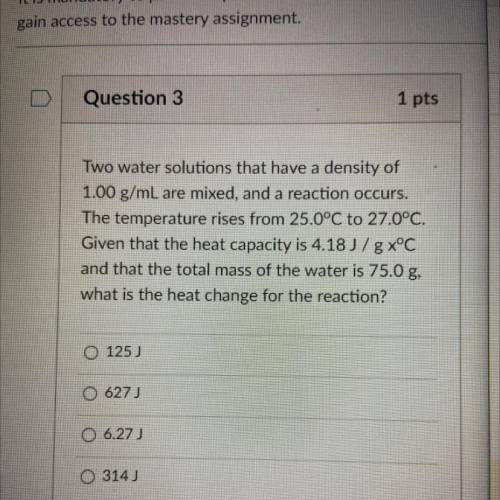
Two water solutions that have a density of
1.00 g/mL are mixed, and a reaction occurs.
The temperature rises from 25.0°C to 27.0°C.
Given that the heat capacity is 4.18 J/g x°C
and that the total mass of the water is 75.0 g,
what is the heat change for the reaction?

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 02:00
What is the source of continuous heat and energy that we receive from the sun
Answers: 2

Chemistry, 23.06.2019 07:40
Which of the following has expanded our knowledge of the universe beyond our solar system the most? a. manned space travel b. the hubble space telescope c. the pioneer and voyager missions d. the international space station
Answers: 3

Chemistry, 23.06.2019 08:00
How does the digestive system interact with the circulatory system? a. messages sent as electrical impulses from the digestive system are transported throughout the body by the circulatory system. b. nutrients taken in and broken down by the digestive system are carried to various parts of the body by the circulatory system. c. nutrients and gases are absorbed by organs in the circulatory system. then, they are transported to all parts of the body by organs in the digestive system. d. oxygen and carbon dioxide are exchanged by organs in the digestive system, and the gases are carried to the rest of the body by the circulatory system.
Answers: 2

Chemistry, 23.06.2019 10:00
How many grams of cupric sulfate pentahydrate are needed to prepare 50.00 ml of 0.0800m cuso4× 5h2o?
Answers: 3
You know the right answer?
Questions

Biology, 12.04.2021 19:20





Biology, 12.04.2021 19:20


Mathematics, 12.04.2021 19:20


Mathematics, 12.04.2021 19:20

Mathematics, 12.04.2021 19:20


Mathematics, 12.04.2021 19:20



Computers and Technology, 12.04.2021 19:20

Mathematics, 12.04.2021 19:20


Mathematics, 12.04.2021 19:20

Mathematics, 12.04.2021 19:20



