
Chemistry, 24.05.2021 18:20 michaelchavez6959127
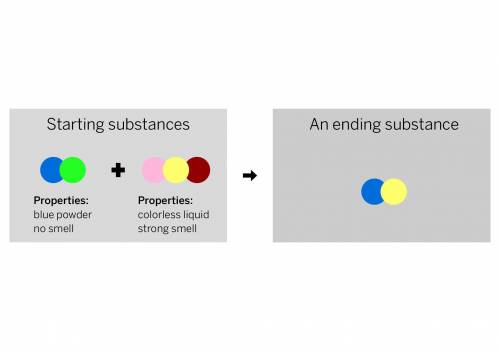
A chemist mixed two substances together: a blue powder with no smell and a colorless liquid with a strong smell. Their repeating groups of atoms are shown above on the left. After they were mixed, the chemist analyzed the results and found two substances. One ending substance had the repeating group of atoms shown above on the right. Is the ending substance the same substance as the blue powder? What happened to the atoms of the starting substances when the ending substances formed? Be sure to explain your answers to both of these questions.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1


Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1

Chemistry, 23.06.2019 00:20
Which diagram represents the phase tha occurs after a solid melts?
Answers: 1
You know the right answer?
A chemist mixed two substances together: a blue powder with no smell and a colorless liquid with a s...
Questions

Mathematics, 15.11.2019 18:31


Mathematics, 15.11.2019 18:31


Spanish, 15.11.2019 18:31

History, 15.11.2019 18:31

English, 15.11.2019 18:31

Social Studies, 15.11.2019 18:31


English, 15.11.2019 18:31


Biology, 15.11.2019 18:31





Biology, 15.11.2019 18:31


Mathematics, 15.11.2019 18:31

Spanish, 15.11.2019 18:31



