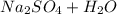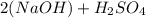Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:30
The ph of carrots are 5.0 how it is classified a.acidic b.basic c.indicator d.neutral
Answers: 2

Chemistry, 22.06.2019 00:30
The clouds are grey and ground is wet. a quantitative b qualitative
Answers: 1


Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3
You know the right answer?
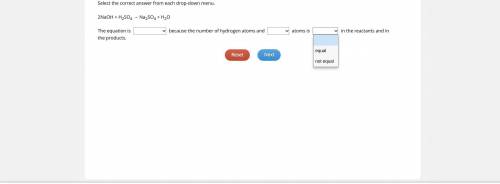
PLEASE HELP
...
...
Questions





Biology, 30.08.2019 05:30


Social Studies, 30.08.2019 05:30




English, 30.08.2019 05:30


Mathematics, 30.08.2019 05:30

Mathematics, 30.08.2019 05:30


Social Studies, 30.08.2019 05:30


Mathematics, 30.08.2019 05:30


Mathematics, 30.08.2019 05:30

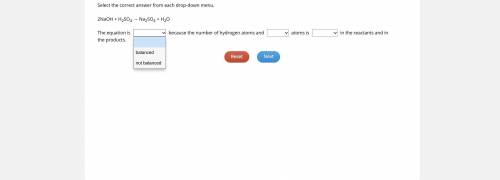
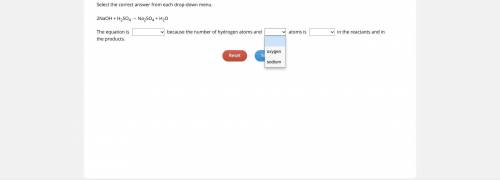
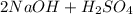 →
→