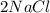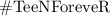Chemistry, 20.05.2021 08:00 mlandon922
Fill in the blanks (Balancing chemical reactions)


Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 05:00
Type the letter that represents the correct location for each particle type below.
Answers: 1

You know the right answer?
Fill in the blanks (Balancing chemical reactions)
...
...
Questions


Mathematics, 07.09.2021 05:10

Mathematics, 07.09.2021 05:10



Mathematics, 07.09.2021 05:10


English, 07.09.2021 05:10


Mathematics, 07.09.2021 05:10

Mathematics, 07.09.2021 05:10


Geography, 07.09.2021 05:10

Business, 07.09.2021 05:10

Mathematics, 07.09.2021 05:10



Mathematics, 07.09.2021 05:10

Physics, 07.09.2021 05:10


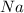 in
in 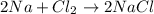 is Coefficient.
is Coefficient. after
after 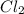 in the same equation is sub script.
in the same equation is sub script.