
Chemistry, 19.05.2021 18:00 jazzyjaz2003
27.5 mL of a 0.235 M potassium hydroxide solution is required to completely react with 35.0 mL of a sulfuric acid solution. Provide the balanced chemical equation for this reaction and determine the concentration of the sulfuric acid solution.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3

Chemistry, 22.06.2019 11:30
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2

Chemistry, 23.06.2019 09:00
What factor besides temperature affects the boiling point of water? a. mass b. number of moles c. volume d. pressure
Answers: 3

Chemistry, 23.06.2019 10:30
Ethyl alcohol, also known as ethanol, has a density of 0.79 g/ml. what is the volume, in quarts, of 1.95 kg of this alcohol?
Answers: 2
You know the right answer?
27.5 mL of a 0.235 M potassium hydroxide solution is required to completely react with 35.0 mL of a...
Questions

Mathematics, 11.12.2020 01:00




Arts, 11.12.2020 01:00




History, 11.12.2020 01:00




Mathematics, 11.12.2020 01:00



Mathematics, 11.12.2020 01:00




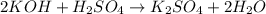 and the concentration of the sulfuric acid solution is 0.184 M.
and the concentration of the sulfuric acid solution is 0.184 M.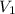 = 27.5 mL,
= 27.5 mL, 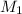 = 0.235 M
= 0.235 M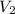 = 35.0 mL,
= 35.0 mL, 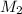 = ?
= ?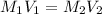
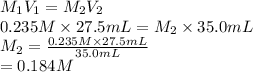

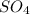 = 1O = 1
= 1O = 1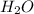 by 2. Hence, the equation can be rewritten as follows.
by 2. Hence, the equation can be rewritten as follows.

