
Chemistry, 19.05.2021 01:00 kaliyaht01
PLEASE HELP ASAP ;(
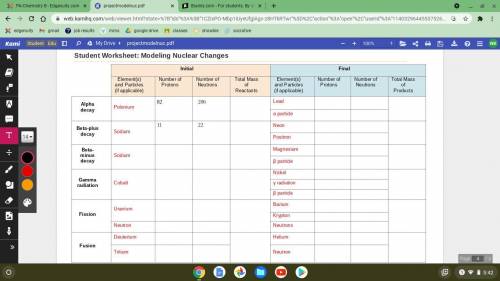
make sure you fill out the table carefully, it is important to recognize that the first four columns are b) for the elements and/or particles at the beginning of the reaction. the four columns on the right are for the final products. be sure your initial total mass is equal to the final total mass, as per the law of conservation of energy.
BASICALLY IF YOU HAVE THIS DONE I NEED HELP W THE ENTIRE PROJECT..

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3


You know the right answer?
PLEASE HELP ASAP ;(
make sure you fill out the table carefully, it is important to recognize that t...
Questions


Physics, 12.02.2021 21:30


Biology, 12.02.2021 21:30

English, 12.02.2021 21:30

Medicine, 12.02.2021 21:30

Mathematics, 12.02.2021 21:30



Mathematics, 12.02.2021 21:30

Mathematics, 12.02.2021 21:30

Chemistry, 12.02.2021 21:30


History, 12.02.2021 21:40

Mathematics, 12.02.2021 21:40

Mathematics, 12.02.2021 21:40

Mathematics, 12.02.2021 21:40

Mathematics, 12.02.2021 21:40

Business, 12.02.2021 21:40

English, 12.02.2021 21:40



