
Chemistry, 18.05.2021 19:30 kmontello5
Equal molar quantities of Ca2 and EDTA (H4Y) are added to make a 0.010 M solution of CaY2- at pH 10. The formation constant for CaY2- is 5.0 x 1010 and the fraction of unprotonated EDTA (Y4-) is 0.35 at pH10. Calculate the concentration of free Ca2 in this solution.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Schrodinger and heisenberg developed an alternate theory about atomic nature that contradicted some of bohr's model of the atom. how do changes resulting from new technology and evidence affect the reputation of the atomic theory?
Answers: 1

Chemistry, 22.06.2019 07:20
2pos suppose an object in free fall is dropped from a building. its starting velocity is 0 m/s. ignoring the effects of air resistance, what is the speed (in m/s) of the object after falling 3 seconds? give your answer as a positive decimal without units. answer here
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 14:00
Calculate the energy required to ionize a hydrogen atom to an excited state where the electron is initially in the n = 5 energy level. report your answer in kilojoules
Answers: 1
You know the right answer?
Equal molar quantities of Ca2 and EDTA (H4Y) are added to make a 0.010 M solution of CaY2- at pH 10....
Questions


Mathematics, 01.06.2021 20:10


Mathematics, 01.06.2021 20:10


Mathematics, 01.06.2021 20:10

Mathematics, 01.06.2021 20:10




Mathematics, 01.06.2021 20:10

Mathematics, 01.06.2021 20:10

Mathematics, 01.06.2021 20:10

Chemistry, 01.06.2021 20:10



Mathematics, 01.06.2021 20:10


Biology, 01.06.2021 20:10

Mathematics, 01.06.2021 20:10

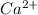 +
+ 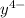 ⇄
⇄ 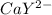
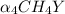 ; ∝₄ = 0.35
; ∝₄ = 0.35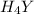 ⇄
⇄ 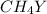 =
= 

