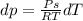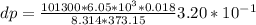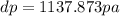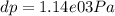Chemistry, 18.05.2021 19:10 chasewilkinson123
Use the fact that to determine how much the pressure must change in order to lower the boiling point of water by a small amount 3.20e-01 K. You may assume that the entropy and density of the liquid and gas are roughly constant for these small changes. You may also assume that the volume per molecule of liquid water is approximately zero compared to that of water vapor, and that water vapor is an ideal gas. Useful constants: Atmospheric pressure is 101300 Pa The boiling point of water at atmospheric pressure is 373.15 K The entropy difference between liquid and gas per kilogram is 6.05e 03 The molecular weight of water is 0.018 kg/mol. (a) 0.00e 00 Pa (b) 1.14e 03 Pa (c) 6.85e 26 Pa (d) 4.24e 05 Pa (e) 3.81e 28 Pa

Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 22:30
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 22.06.2019 00:30
In numbering carbon atoms in the parent chain of a hydrocarbon, why would you number from right to left, rather than left to right
Answers: 1

You know the right answer?
Use the fact that to determine how much the pressure must change in order to lower the boiling point...
Questions

Mathematics, 23.08.2021 17:50





Chemistry, 23.08.2021 17:50

Biology, 23.08.2021 17:50



Mathematics, 23.08.2021 17:50

Mathematics, 23.08.2021 17:50





History, 23.08.2021 17:50


English, 23.08.2021 17:50


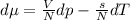 to determine how much the pressure must change in order to lower the boiling point of water by a small amount 3.20e-01 K. You may assume that the entropy and density of the liquid and gas are roughly constant for these small changes. You may also assume that the volume per molecule of liquid water is approximately zero compared to that of water vapor, and that water vapor is an ideal gas. Useful constants: Atmospheric pressure is 101300 Pa The boiling point of water at atmospheric pressure is 373.15 K The entropy difference between liquid and gas per kilogram is 6.05e 03 J/kgK The molecular weight of water is 0.018 kg/mol. (a) 0.00e 00 Pa (b) 1.14e 03 Pa (c) 6.85e 26 Pa (d) 4.24e 05 Pa (e) 3.81e 28 Pa
to determine how much the pressure must change in order to lower the boiling point of water by a small amount 3.20e-01 K. You may assume that the entropy and density of the liquid and gas are roughly constant for these small changes. You may also assume that the volume per molecule of liquid water is approximately zero compared to that of water vapor, and that water vapor is an ideal gas. Useful constants: Atmospheric pressure is 101300 Pa The boiling point of water at atmospheric pressure is 373.15 K The entropy difference between liquid and gas per kilogram is 6.05e 03 J/kgK The molecular weight of water is 0.018 kg/mol. (a) 0.00e 00 Pa (b) 1.14e 03 Pa (c) 6.85e 26 Pa (d) 4.24e 05 Pa (e) 3.81e 28 Pa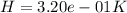
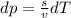
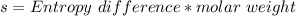
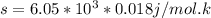
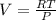 (from ideal gas equation)
(from ideal gas equation)