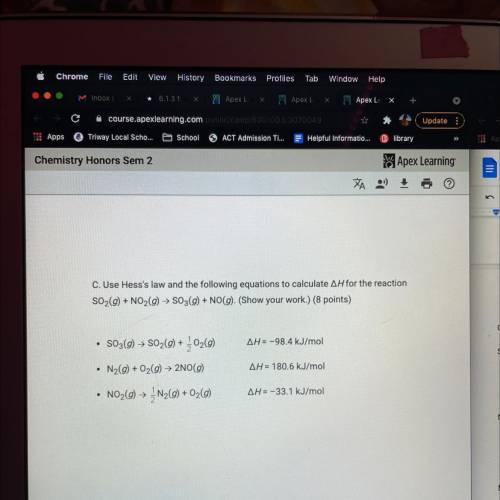
Chem exam ap3x
Look at the attached image for the questions
...


Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 21:30
In one or two grammatically correct sentences, write a definition for the term molecule geometry
Answers: 3

Chemistry, 22.06.2019 22:00
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2

Chemistry, 23.06.2019 02:30
When the ionic compound nabr dissolves in water, br– ions are pulled into solution by the attraction between what two particles? a. the na+ and br– ions b. the na+ ion and the negative end of a water molecule c. the br– ion and the positive end of a water molecule d. the br– ion and the negative end of a water molecule
Answers: 1
You know the right answer?
Questions

Mathematics, 30.06.2019 12:30


History, 30.06.2019 12:30

Biology, 30.06.2019 12:30

Physics, 30.06.2019 12:30


Social Studies, 30.06.2019 12:30

Social Studies, 30.06.2019 12:30

Mathematics, 30.06.2019 12:30

Social Studies, 30.06.2019 12:30

Mathematics, 30.06.2019 12:30

Health, 30.06.2019 12:30

Biology, 30.06.2019 12:30

Mathematics, 30.06.2019 12:30

Mathematics, 30.06.2019 12:30



Business, 30.06.2019 12:30

Social Studies, 30.06.2019 12:30

Geography, 30.06.2019 12:30