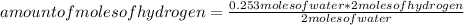Chemistry, 17.05.2021 17:50 aide1234564
The balanced chemical equation for the reaction of hydrogen and oxygen is shown below. 2H2 + O2 Right arrow. 2H2O The number of moles of hydrogen that is needed to produce 0.253 mol of water is

Answers: 1


Another question on Chemistry

Chemistry, 20.06.2019 18:04
The volume of a sphere is given by v= (4/3) pi r cubed, where r is the radius. compute the volume of a sphere with a radius of 117pm. state your answer in units of cubed.
Answers: 1



Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2
You know the right answer?
The balanced chemical equation for the reaction of hydrogen and oxygen is shown below. 2H2 + O2 Righ...
Questions

Geography, 10.01.2020 12:31



Chemistry, 10.01.2020 12:31

Biology, 10.01.2020 12:31



Biology, 10.01.2020 12:31

Mathematics, 10.01.2020 12:31




Biology, 10.01.2020 12:31

Mathematics, 10.01.2020 12:31

Mathematics, 10.01.2020 12:31


Mathematics, 10.01.2020 12:31

Mathematics, 10.01.2020 12:31

Mathematics, 10.01.2020 12:31

History, 10.01.2020 12:31