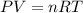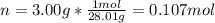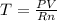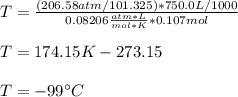Chemistry, 17.05.2021 16:50 colyernicholas44
A sample of nitrogen gas in a 750.0 mL sealed flask exerts a pressure of 206.58 kPa. Calculate the temperature of the gas if the flask contains 3.00 grams of gas.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Your answer should have the same number or significant figures as a he starting measurement. 3201 ml =
Answers: 2

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 23.06.2019 00:30
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2

You know the right answer?
A sample of nitrogen gas in a 750.0 mL sealed flask exerts a pressure of 206.58 kPa. Calculate the t...
Questions



Physics, 21.06.2019 16:10



Mathematics, 21.06.2019 16:10

Mathematics, 21.06.2019 16:10

Mathematics, 21.06.2019 16:10

Mathematics, 21.06.2019 16:10


Mathematics, 21.06.2019 16:10