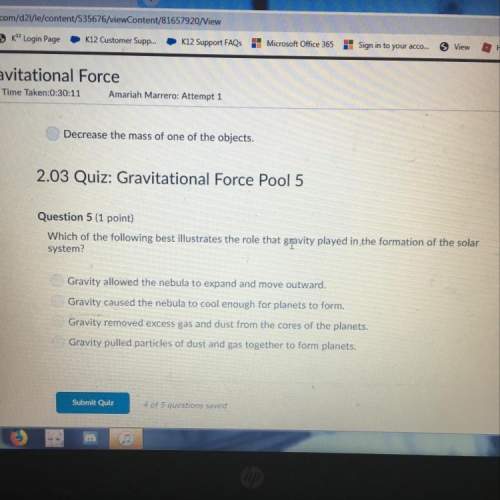
A 72.0-gram piece of metal at 96.0 °C is placed in 130.0 g of water in a calorimeter at 25.5 °C. The final temperature in the calorimeter is 31.0 °C. Determine the specific heat of the metal. Show your work by listing various steps, and explain how the law of conservation of energy applies to this situation. (10 points)

Answers: 3


Another question on Chemistry


Chemistry, 21.06.2019 20:30
When curium-242 is bombarded with an alpha particle, two products are formed, one of which is a nudge on. what is the other product
Answers: 3

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 08:30
How would the number of moles (n) of o2 change if the atmospheric pressure doubled but all other variables stayed the same
Answers: 2
You know the right answer?
A 72.0-gram piece of metal at 96.0 °C is placed in 130.0 g of water in a calorimeter at 25.5 °C. The...
Questions




History, 12.11.2019 23:31

Mathematics, 12.11.2019 23:31

Mathematics, 12.11.2019 23:31


Spanish, 12.11.2019 23:31



Geography, 12.11.2019 23:31


Mathematics, 12.11.2019 23:31

Mathematics, 12.11.2019 23:31


Mathematics, 12.11.2019 23:31