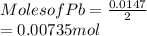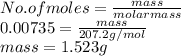When a lead acid car battery is recharged by the alternator, it acts essentially as an electrolytic cell in which solid lead(II) sulfate PbSO4 is reduced to lead at the cathode and oxidized to solid lead(II) oxide PbO at the anode. Suppose a current of 62.0 is fed into a car battery for 23.0 seconds. Calculate the mass of lead deposited on the cathode of the battery. Round your answer to 3 significant digits. Also, be sure your answer contains a unit symbol.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1


Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 23.06.2019 06:10
How can liquids be seperated by density a the liquids are absorbed onto a paper b the liquids are turned into seperate vapors c the liquids are collected as they evaporate d the liquids are allowed to seperate into layers
Answers: 1
You know the right answer?
When a lead acid car battery is recharged by the alternator, it acts essentially as an electrolytic...
Questions


Mathematics, 18.02.2020 23:19






English, 18.02.2020 23:20



Mathematics, 18.02.2020 23:20

Mathematics, 18.02.2020 23:21





Physics, 18.02.2020 23:21



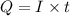
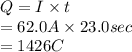
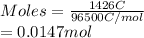
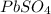 is 2. So, moles deposited by Pb is as follows.
is 2. So, moles deposited by Pb is as follows.