Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3
You know the right answer?
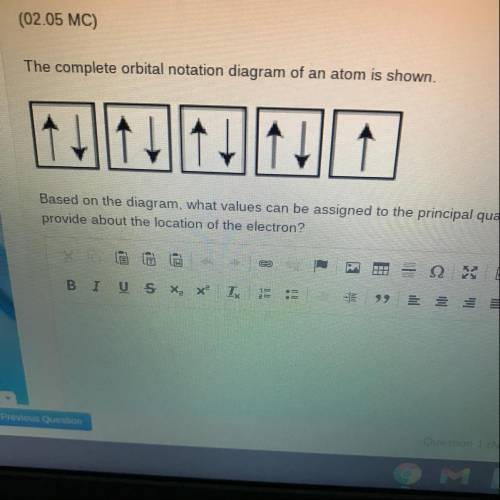
The complete orbital notation diagram of an atom is shown
Based on the diagram, what values can be...
Questions



Mathematics, 09.07.2019 13:30

English, 09.07.2019 13:30

Mathematics, 09.07.2019 13:30

Chemistry, 09.07.2019 13:30

Mathematics, 09.07.2019 13:30

Computers and Technology, 09.07.2019 13:30

Biology, 09.07.2019 13:30


Chemistry, 09.07.2019 13:30

Social Studies, 09.07.2019 13:30


History, 09.07.2019 13:30




History, 09.07.2019 13:30


English, 09.07.2019 13:30