
Chemistry, 14.05.2021 20:10 phanuel642
to whoever answers correctly first!
(I didn't know what physical science fell under so I picked a random science related subject)
Dominic made the table below to organize his notes about mixtures.
A 1-column table.
The first column labeled properties of mixtures has entries has no set composition, must have more than one state of matter, must have more than one substance.
What mistake did Dominic make?
The title should read “Properties of Solutions” because some mixtures do not have all of the properties listed.
There is a definite recipe to make each mixture, so the composition of a mixture is set. Although it is possible to have more than one state, it is also possible to have only one state.
A single substance can be used to make a mixture if the substance is composed of more than one element.
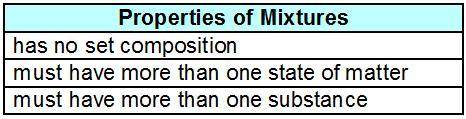

Answers: 2


Another question on Chemistry



Chemistry, 23.06.2019 04:10
Two solids are mixed in a flask and stirred. after a few minutes, the flask becomes cold. which of the following best describes this reaction? a. an exothermic reaction b. a combustion reaction c. an endothermic reaction d. a decomposition reaction
Answers: 1

Chemistry, 23.06.2019 06:40
1.) which of the following is a molecule but not a compound? a.he b.f2 c.h2o d.ch4 2.) what is a physical combination of substances? a.a compound b.a molecule c.a mixture d.an element 3.) what is a chemical combination of substances? a.a compound b.an atom c.a mixture d.an element 4.) what is the relationship between the solute and solvent in a solution? a.they form a compound b.they form a mixture c.they form molecules d.they form chemical bonds 5.) the gases in air dissolve in water. what would be one way to reduce the amount of a gas dissolved in water? a.add more water b.reduce the air pressure c.increase the air pressure d.stir the water 6.) how would you determine the solubility of a substance? a.find how well it dissolved various substances. b.find the mass and the volume of the substance. c.find the temperature at which the substance evaporated. d.find how much i was able to dissolve in a solute. 7.) the periodic table organizes all of the kinds of a.molecules. b.compounds. c.atoms. d.ions. 8.)what distinguishes two substances combined to become a compound vs. two substances combined to become a mixture? a.whether they can be easily separated b.whether they chemically bond together c.whether they both are visible d.whether they are heterogeneous 9.) the principle components of air are: n2 78% o2 21% ar 0.95% co2 0.038% this is a solution of a.molecules and atoms. b.molecules. c.compounds and molecules. d.atoms.
Answers: 1
You know the right answer?
to whoever answers correctly first!
(I didn't know what physical science fell under so I picked a r...
Questions











Mathematics, 27.05.2021 22:50





Mathematics, 27.05.2021 22:50


Mathematics, 27.05.2021 22:50




