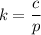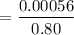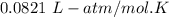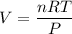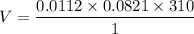The solubility of N2in blood at 37°C and at a partial pressure of 0.80 atm is 5.6 × 10−4mol/L. A deep-sea diver breathes compressed air with the partial pressure of N2equal to 4.0 atm. Assume that the total volume of blood in the body is 5.0 L. Calculate the amount of N2gas released (in liters at 37°C and 1 atm) when the diver returns to the surface of the water, where the partial pressure ofN2is 0.80 atm.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
This graph gives information on changes in sea ice extent in the arctic ocean over a 30-year span. the overall trend shows in the ice extent. to address the trend, scientists need to ask themselves, one direct consequence of the trend is that
Answers: 1

Chemistry, 22.06.2019 00:40
1) in saturated limewater, [h+ ]=3.98x10-13 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 2) in butter, [h+ ]=6.0x10-7 m. a) find [oh]-/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 3) in peaches, [oh]=3.16x10-11 m a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? / 4) during the course of the day, human saliva varies between being acidic and basic. if [oh]=3.16x10-8 m, a) find [h+ ]/ b) what is the ph? / c) is the solution acidic, basic, or neutral? /
Answers: 3


Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1
You know the right answer?
The solubility of N2in blood at 37°C and at a partial pressure of 0.80 atm is 5.6 × 10−4mol/L. A dee...
Questions


Mathematics, 04.12.2020 23:30


Physics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Geography, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30

Mathematics, 04.12.2020 23:30




English, 04.12.2020 23:30

Chemistry, 04.12.2020 23:30

Computers and Technology, 04.12.2020 23:30



Mathematics, 04.12.2020 23:30