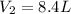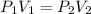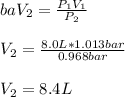Chemistry, 14.05.2021 15:30 joshawajackson
A balloon is inflated to a volume of 8.0 L on a day when the atmospheric pressure is 1.013 bar . The next day, a storm front arrives, and the atmospheric pressure drops to 0.968 bar . Assuming the temperature remains constant, what is the new volume of the balloon, in liters

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 04:30
Using the periodic table, complete the table to describe each atom. type in your answers
Answers: 3

Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 22.06.2019 19:30
What is the area in square meters of 448 g ai foil that has a thickness of 23921 nm? the density is 2.70 g/cm
Answers: 3
You know the right answer?
A balloon is inflated to a volume of 8.0 L on a day when the atmospheric pressure is 1.013 bar . The...
Questions

Arts, 16.12.2021 23:30



Mathematics, 16.12.2021 23:30







Health, 16.12.2021 23:30


Mathematics, 16.12.2021 23:30



Mathematics, 16.12.2021 23:30


Chemistry, 16.12.2021 23:30

Mathematics, 16.12.2021 23:30