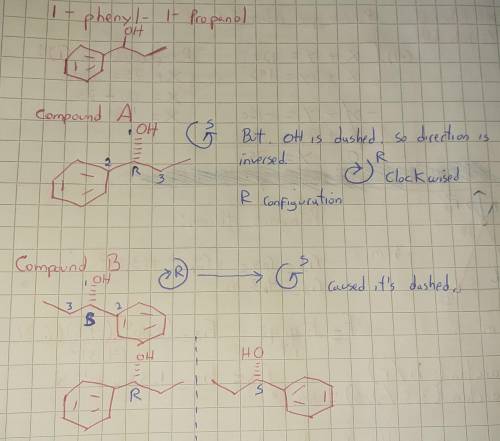
Each of the given molecules contains a chirality center. Determine the relationship between the molecules. three images of 1-phenyl-1-propanol are given as compound A, B and C. Compound A has a dashed bond to the alcohol group, with the phenyl group on the left side and the remaining carbon chain on the right. Compound B has a dashed bond to the alcohol group, with the phenyl group on the right side and the remaining carbon chain on the left. Compound C has a wedge bond to the alcohol group, with the phenyl group on the right side and the remaining carbon chain on the left. Compound D is 1-phenyl-2-propanol with a dashed bond to the alcohol group and the phenyl on the left side and the remaining single carbon on the right side. What is the relationship between A and B

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:00
What is the theoretical yield of carbon dioxide? a)0.993 gb)2.98 gc)3.65 gd)8.93 g
Answers: 1

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 23.06.2019 03:30
Name 3 types of energy you see being used as you look around a classroom
Answers: 1
You know the right answer?
Each of the given molecules contains a chirality center. Determine the relationship between the mole...
Questions

Mathematics, 02.12.2020 19:20


Social Studies, 02.12.2020 19:20

Mathematics, 02.12.2020 19:20

English, 02.12.2020 19:20

Chemistry, 02.12.2020 19:20


Mathematics, 02.12.2020 19:20





Mathematics, 02.12.2020 19:20



Biology, 02.12.2020 19:20

Mathematics, 02.12.2020 19:20


Mathematics, 02.12.2020 19:20