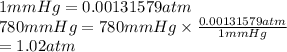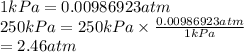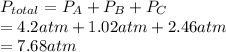Chemistry, 12.05.2021 16:00 VanBrocklin8501
Find the total pressure (in atm) of a mixture that contains 3 gases with the following partial pressures. Gas A is 4.2atm, Gas B is 780 mmHg, Gas C is 250.00kPa

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2

Chemistry, 23.06.2019 00:30
What is the percent by mass of magnesium sulfate in mgso4.7h2o
Answers: 3
You know the right answer?
Find the total pressure (in atm) of a mixture that contains 3 gases with the following partial press...
Questions



Computers and Technology, 07.09.2021 18:40




Mathematics, 07.09.2021 18:40



Biology, 07.09.2021 18:40


Mathematics, 07.09.2021 18:40



Physics, 07.09.2021 18:40


English, 07.09.2021 18:40



Social Studies, 07.09.2021 18:40