
An 18.0 g sample of unidentified metal was heated from 21.5°C to 89.0°C.
292 J of heat energy was absorbed by the metal in the heating process.
Calculate the specific heat of the metal to determine its identity.(need work show, or how you got the answer)
A. calcium
B. iron
C. copper
D. silver
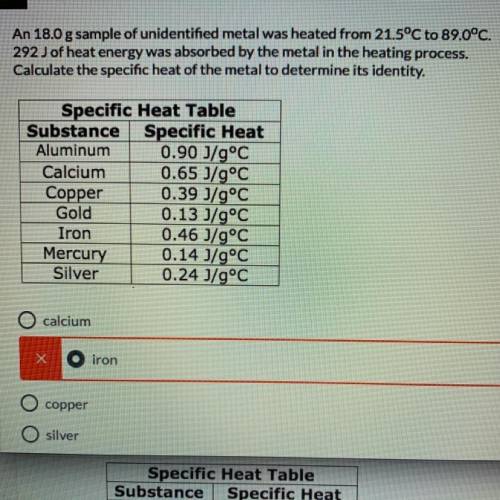

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 3
You know the right answer?
An 18.0 g sample of unidentified metal was heated from 21.5°C to 89.0°C.
292 J of heat energy was a...
Questions

Physics, 17.01.2020 10:31

English, 17.01.2020 10:31

History, 17.01.2020 10:31

History, 17.01.2020 10:31

Biology, 17.01.2020 10:31



Mathematics, 17.01.2020 10:31

English, 17.01.2020 10:31







Mathematics, 17.01.2020 10:31

History, 17.01.2020 10:31



Biology, 17.01.2020 10:31

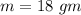
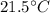 to
to 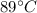
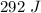
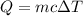
![\Rightarrow 292=18\times c\times [89-21.5]\\\Rightarrow 292=18\times c\times 67.5\\\\\Rightarrow c=\dfrac{292}{1215}\\\\\Rightarrow c=0.24\ J/g^{\circ}C](/tpl/images/1318/5230/23712.png)


