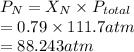Chemistry, 12.05.2021 04:30 irabrooks14
To lower the chance of suffering from decompression sickness (the bends), scuba divers use a mixture of gases in their air tank (typically oxygen and nitrogen gas in recreational dives). Assuming no other gas is present besides oxygen and nitrogen, if the mole fraction of oxygen present is 0.21, what is the partial pressure of nitrogen gas if the total pressure is 111.7 atm

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2
You know the right answer?
To lower the chance of suffering from decompression sickness (the bends), scuba divers use a mixture...
Questions

Chemistry, 20.09.2020 03:01

Chemistry, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01


Biology, 20.09.2020 03:01




English, 20.09.2020 03:01


English, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01


Mathematics, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01


Mathematics, 20.09.2020 03:01


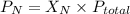
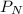 = partial pressure of nitrogen
= partial pressure of nitrogen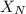 = mole fraction of nitrogen
= mole fraction of nitrogen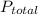 = total pressure
= total pressure