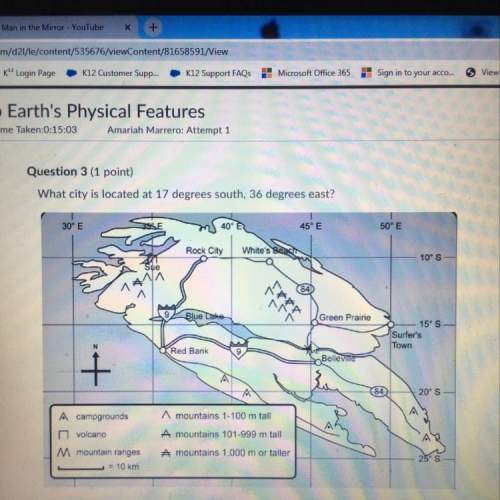
You are conducting a calorimetry experiment using a calorimeter with 500 mL of water in its outer
chamber to determine the enthalpy of reaction of a chemical reaction. The initial temperature of the
water was 25 °C. After the reaction, the temperature of the water was 57.4 °C. Disregarding any heat
loss to the walls of the container, calculate the Hf). (The specific heat of the water is 4.18 J/(g. K).

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 05:30
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease.correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1


Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1
You know the right answer?
You are conducting a calorimetry experiment using a calorimeter with 500 mL of water in its outer
c...
Questions


English, 29.10.2019 17:31


History, 29.10.2019 17:31

History, 29.10.2019 17:31




Mathematics, 29.10.2019 17:31

Mathematics, 29.10.2019 17:31

English, 29.10.2019 17:31




Advanced Placement (AP), 29.10.2019 17:31

History, 29.10.2019 17:31


Mathematics, 29.10.2019 17:31

Mathematics, 29.10.2019 17:31