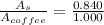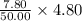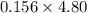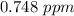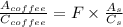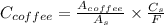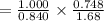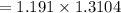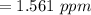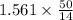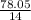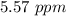Chemistry, 11.05.2021 18:40 haloom9698
A sample was then prepared containing 14.00 mL of coffee and 8.00 mL of 4.80 ppm Li , and diluted to a 50.00 mL total volume. The ratio of the internal standard signal to the analyte signal in the unknown sample was found to be 0.840/1.000 (Li signal/Na signal). Determine the unknown concentration of Na in the coffee sample.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3


Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1

Chemistry, 23.06.2019 03:00
What happens in the particles of a gas when the gas is compressed
Answers: 1
You know the right answer?
A sample was then prepared containing 14.00 mL of coffee and 8.00 mL of 4.80 ppm Li , and diluted to...
Questions

Biology, 04.01.2021 04:30

Biology, 04.01.2021 04:30




History, 04.01.2021 04:40

Mathematics, 04.01.2021 04:40



Mathematics, 04.01.2021 04:40

Mathematics, 04.01.2021 04:40

French, 04.01.2021 04:40



Business, 04.01.2021 04:40

Engineering, 04.01.2021 04:40

Mathematics, 04.01.2021 04:40



Mathematics, 04.01.2021 04:40

![[Na^+]=5.57 \ ppm](/tpl/images/1316/5042/79840.png) ".
".