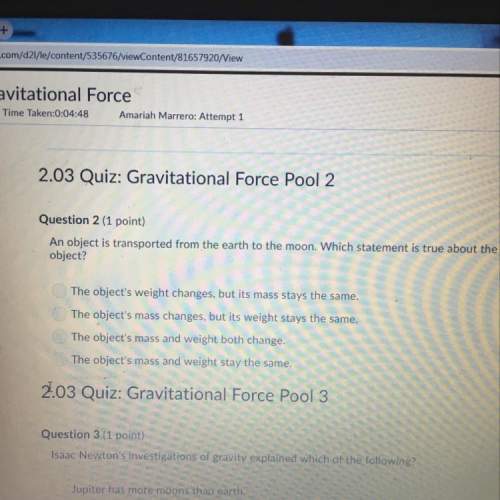
Question 23 (1 point)
The theoretical yield of water that can be produced by reacting hydrogen and oxygen is 0.47g. After doing the lab, the actual
yield of water produced was 0.35g. What is the percent yield of water that was produced?
O a
74.47%
134.28%
Ob
Ос
1.3496
Od
0.7496

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 2

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1
You know the right answer?
Question 23 (1 point)
The theoretical yield of water that can be produced by reacting hydrogen and...
Questions


Arts, 17.02.2021 18:10

Mathematics, 17.02.2021 18:10



Biology, 17.02.2021 18:10

Mathematics, 17.02.2021 18:10

English, 17.02.2021 18:10

Mathematics, 17.02.2021 18:10

Arts, 17.02.2021 18:10


Mathematics, 17.02.2021 18:10


English, 17.02.2021 18:10

Mathematics, 17.02.2021 18:10



Computers and Technology, 17.02.2021 18:10

History, 17.02.2021 18:10

Mathematics, 17.02.2021 18:10