
Chemistry, 07.05.2021 05:30 anniekwilbourne
POSSIBLE
(1-6) Which statement best describes the energy involved when an electron goes from shell 3 to shell 1 as shown in picture below.
1: Light energy is being absorbed
2: Heat energy is being emitted
3: Light energy is being emitted
4: Heat energy is being absorbed
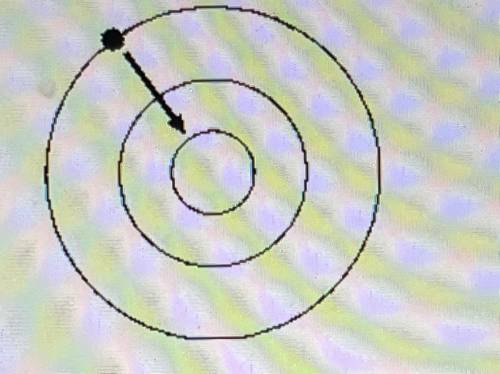

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3

Chemistry, 23.06.2019 04:20
Calculate the mass of 0.750 mol of the following substance. na3po4.
Answers: 1
You know the right answer?
POSSIBLE
(1-6) Which statement best describes the energy involved when an electron goes from shell...
Questions


Social Studies, 17.10.2019 09:50

Chemistry, 17.10.2019 09:50


History, 17.10.2019 09:50


Mathematics, 17.10.2019 09:50


English, 17.10.2019 09:50

Mathematics, 17.10.2019 09:50

English, 17.10.2019 09:50

History, 17.10.2019 09:50


History, 17.10.2019 09:50

History, 17.10.2019 09:50


Chemistry, 17.10.2019 09:50


Mathematics, 17.10.2019 09:50



