
Chemistry, 07.05.2021 04:10 krystalhurst97
Calculations:
Show your calculations for each of the following. Remember, calculations should follow rules for significant figures.
Write the balanced chemical equation for the reaction you are performing.
Mg(s) + O2(g) → MgO(s)
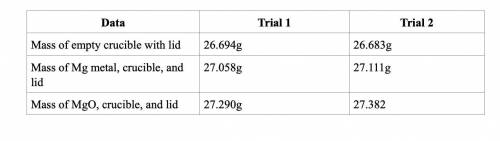
Subtract the mass of the crucible and lid (row 1 in the chart) from the total mass of Mg, crucible, and lid (row 2 in the chart) to find the mass of magnesium for each trial.
Trial 1: 0.364
Trial 2: 0.428
Subtract the mass of the crucible and lid (row 1 in the chart) from the total mass of MgO, crucible, and lid (row 3 in the chart) to find the mass of magnesium oxide for each trial. This is the actual yield of magnesium oxide for each trial.
Trial 1: 0.596
Trial 2: 0.699
Magnesium is the limiting reactant in this experiment. Calculate the theoretical yield of MgO for each trial.
Trial 1:
Trial 2:
Determine the percent yield of MgO for your experiment for each trial.
Trial 1:
Trial 2:
Determine the average percent yield of MgO for the two trials.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:10
Which statement describes both homogeneous mixtures and heterogeneous mixtures?
Answers: 1

Chemistry, 22.06.2019 08:00
If 90.0 grams of ethane reacted with excess chlorine,how many grams of dicarbon hexachloride would form
Answers: 1

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3
You know the right answer?
Calculations:
Show your calculations for each of the following. Remember, calculations should foll...
Questions


Geography, 17.06.2021 18:40

Mathematics, 17.06.2021 18:40



History, 17.06.2021 18:40



English, 17.06.2021 18:40


Computers and Technology, 17.06.2021 18:40


Computers and Technology, 17.06.2021 18:40




Mathematics, 17.06.2021 18:40





