
Chemistry, 05.05.2021 23:20 petroale000
Calculate the enthalpy change in kJ/mol for the reaction: C3H8(g) + 5O2(g) ---> 3CO2(g) + 4H2O(g)
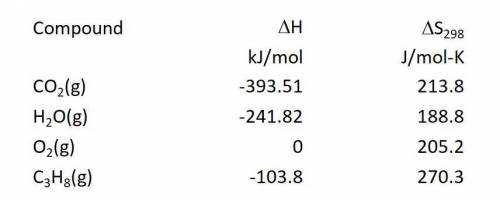

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 07:30
All cells are made of four types of acids, lipids, proteins, and carbohydrates.
Answers: 1

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 17:10
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1
You know the right answer?
Calculate the enthalpy change in kJ/mol for the reaction:
C3H8(g) + 5O2(g) ---> 3CO2(g) + 4H2O(...
Questions


History, 29.08.2019 18:00



Mathematics, 29.08.2019 18:00


Mathematics, 29.08.2019 18:00

Mathematics, 29.08.2019 18:00

History, 29.08.2019 18:00


Biology, 29.08.2019 18:00



Mathematics, 29.08.2019 18:00



Mathematics, 29.08.2019 18:00

History, 29.08.2019 18:00

Social Studies, 29.08.2019 18:00

Biology, 29.08.2019 18:00



