
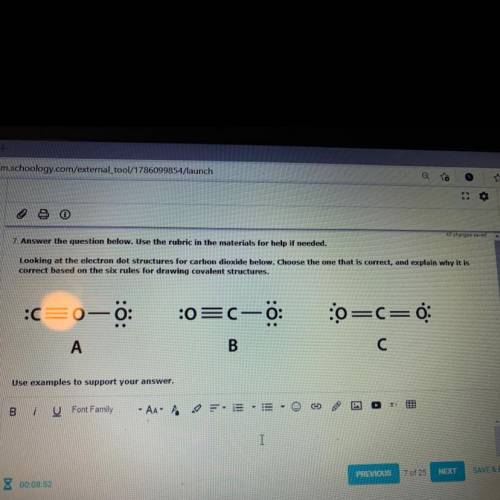
7. Answer the question below. Use the rubric in the materials for help if needed.
Looking at the electron dot structures for carbon dioxide below. Choose the one that is correct, and explain why it is
correct based on the six rules for drawing covalent structures.
:c=0-0
:0=C—0:
:0=C=0
A
B
C
Ilse eyamnles to support your answer.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Astudent is given a sample of a blue copper sulfate hydrate. he weighs the sample in a dry covered porcelain crucible and got a mass of 23.875 g for the crucible, lid, and sample. the mass of the empty crucible and lid was found earlier to be 22.652 g. he then heats the crucible to expel the water of hydration, keeping the crucible at red heat for 10 minutes with the lid slightly ajar. on colling, he finds the mass of crucible, lid, and contents to be 23.403 g. the sample was changed in the process to very light clue anhydrous cuso4. if there are again 100.0 g of hydrate, how many grams of cuso4 are in it? how many moles of cuso4? (hint: molar mass of cuso4 = 159.6 g / mole. what per cent of the hydrate is cuso4? you may convert the mass of cuso4 to moles.)
Answers: 3

Chemistry, 22.06.2019 04:40
In which environment would primary succession occur? a forest with a few remaining trees after a recent wildfire an area of exposed rock after a glacier melts away beach that is exposed to the air at low tide an abandoned baseball field in a small town
Answers: 1

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1
You know the right answer?
7. Answer the question below. Use the rubric in the materials for help if needed.
Looking at the e...
Questions



Mathematics, 31.03.2021 06:40

Mathematics, 31.03.2021 06:40


Mathematics, 31.03.2021 06:40

Mathematics, 31.03.2021 06:40


Social Studies, 31.03.2021 06:40

English, 31.03.2021 06:40

Mathematics, 31.03.2021 06:40


Mathematics, 31.03.2021 06:40

History, 31.03.2021 06:40





Advanced Placement (AP), 31.03.2021 06:40

Mathematics, 31.03.2021 06:40



