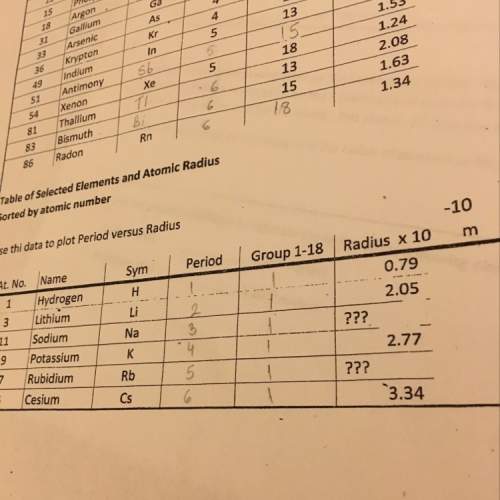
Chemistry, 29.04.2021 19:10 niceguy1997
Na and HCl are colliding as shown below with an energy
Q1. Will this collision be effective? Explain
Q2. What are 2 things you could change to increase the rate of this reaction? Use
the following words to explain your number of collision, and kinetic energy.
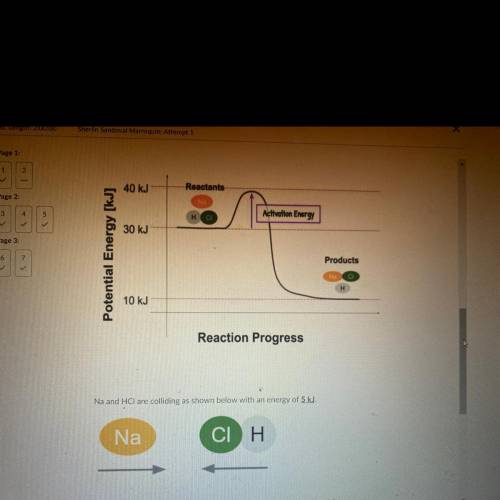

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2

Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1

Chemistry, 22.06.2019 18:00
Answer asap need to be answered by wednesday morning explain how a buffer works, using an ethanoic acid / sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 3

Chemistry, 22.06.2019 19:50
What is the wavelength of a wave with a velocity of 50 m/s and a frequency of 5hz a 250 m b 0.1 m c 10m d 0.01 m
Answers: 2
You know the right answer?
Na and HCl are colliding as shown below with an energy
Q1. Will this collision be effective? Expla...
Questions


Computers and Technology, 01.12.2021 22:50


Mathematics, 01.12.2021 22:50



Mathematics, 01.12.2021 22:50


History, 01.12.2021 22:50

Mathematics, 01.12.2021 22:50

Mathematics, 01.12.2021 22:50


Mathematics, 01.12.2021 22:50

Physics, 01.12.2021 22:50

History, 01.12.2021 22:50

History, 01.12.2021 22:50