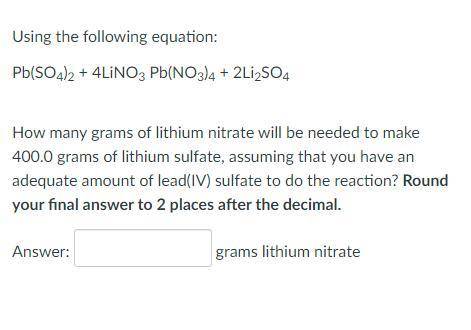
Using the following equation:
Pb(SO4)2 + 4LiNO3 Pb(NO3)4 + 2Li2SO4
How many grams of l...

Chemistry, 27.04.2021 18:20 LilLappyLOL
Using the following equation:
Pb(SO4)2 + 4LiNO3 Pb(NO3)4 + 2Li2SO4
How many grams of lithium nitrate will be needed to make 400.0 grams of lithium sulfate, assuming that you have an adequate amount of lead(IV) sulfate to do the reaction? Round your final answer to 2 places after the decimal.
_ grams lithium nitrate

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
If the particles in a sample of matter have an orderly arrangement and move only in place, the sample is a
Answers: 1

Chemistry, 22.06.2019 11:30
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3

Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

You know the right answer?
Questions

Mathematics, 29.12.2019 04:31






Computers and Technology, 29.12.2019 04:31


Social Studies, 29.12.2019 04:31



Mathematics, 29.12.2019 04:31



Geography, 29.12.2019 04:31


History, 29.12.2019 04:31


Social Studies, 29.12.2019 04:31



