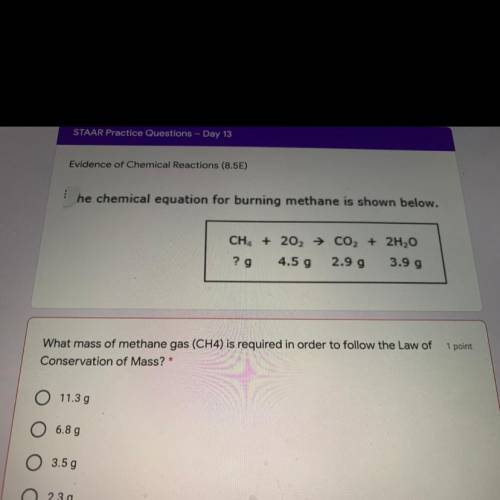
What mass of methane gas (CH4) is required in order to follow the Law of
Conservation of Mass?...

Chemistry, 27.04.2021 17:40 haileysolis5
What mass of methane gas (CH4) is required in order to follow the Law of
Conservation of Mass?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 02:10
Determine the percent sulfuric acid by mass of a 1.61 m aqueous solution of h2so4. %
Answers: 2

Chemistry, 22.06.2019 04:40
In which environment would primary succession occur? a forest with a few remaining trees after a recent wildfire an area of exposed rock after a glacier melts away beach that is exposed to the air at low tide an abandoned baseball field in a small town
Answers: 1

You know the right answer?
Questions

Physics, 03.03.2020 19:59

Biology, 03.03.2020 19:59


Geography, 03.03.2020 19:59


History, 03.03.2020 19:59

Mathematics, 03.03.2020 19:59


Mathematics, 03.03.2020 19:59

Computers and Technology, 03.03.2020 19:59

English, 03.03.2020 19:59











