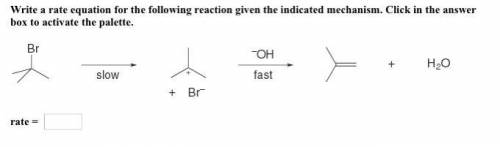
Please don't send any links, please
...

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2

You know the right answer?
Questions

English, 14.08.2021 01:40

Mathematics, 14.08.2021 01:40

Chemistry, 14.08.2021 01:40

Mathematics, 14.08.2021 01:40






Mathematics, 14.08.2021 01:40






Mathematics, 14.08.2021 01:40

Mathematics, 14.08.2021 01:40

Mathematics, 14.08.2021 01:40

Computers and Technology, 14.08.2021 01:50

Computers and Technology, 14.08.2021 01:50