Identify each of the following as a single bond, double bond, or triple bond.
...

Chemistry, 21.04.2021 19:00 SenpaiTaii
Identify each of the following as a single bond, double bond, or triple bond.
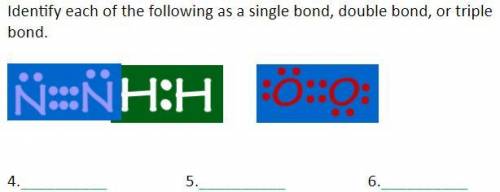

Answers: 3


Another question on Chemistry


Chemistry, 23.06.2019 01:30
Select the correct answer from each drop-down menu. to make a table of the elements, dmitri mendeleev sorted the elements according to their . he then split the list of elements into several columns so that elements beside each other had similar .
Answers: 2

Chemistry, 23.06.2019 06:10
How can liquids be seperated by density a the liquids are absorbed onto a paper b the liquids are turned into seperate vapors c the liquids are collected as they evaporate d the liquids are allowed to seperate into layers
Answers: 1

Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2
You know the right answer?
Questions

Mathematics, 23.10.2020 18:20

History, 23.10.2020 18:20

SAT, 23.10.2020 18:20

Mathematics, 23.10.2020 18:20

Advanced Placement (AP), 23.10.2020 18:20


English, 23.10.2020 18:20

Mathematics, 23.10.2020 18:20

Mathematics, 23.10.2020 18:20

Mathematics, 23.10.2020 18:20


Chemistry, 23.10.2020 18:20



Biology, 23.10.2020 18:20

Mathematics, 23.10.2020 18:20


Social Studies, 23.10.2020 18:20


Mathematics, 23.10.2020 18:20



