
Chemistry, 24.08.2019 23:20 eeromaki1321
For the reaction:
6li(s)+n2(g)→2li3n(s)
determine:
the mass of n2 needed to react with 0.536 moles of li.
the number of moles of li required to make 46.4 g of li3n.
the mass in grams of li3n produced from 3.65 g li.
the number of moles of lithium needed to react with 7.00 grams of n2.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1

Chemistry, 23.06.2019 00:00
How many atoms or molecules are there in a mole of a substance?
Answers: 1

Chemistry, 23.06.2019 07:30
How do you interpret a chromagram for what mixtures contain?
Answers: 1

Chemistry, 23.06.2019 08:30
If you had to research a particular disease or area of concern in veterinary medicine and science, which one would you choose? why?
Answers: 1
You know the right answer?
For the reaction:
6li(s)+n2(g)→2li3n(s)
determine:
the mass of n2 needed to reac...
6li(s)+n2(g)→2li3n(s)
determine:
the mass of n2 needed to reac...
Questions

Mathematics, 07.04.2021 05:10








Chemistry, 07.04.2021 05:10

History, 07.04.2021 05:10

Mathematics, 07.04.2021 05:10

Social Studies, 07.04.2021 05:10

Mathematics, 07.04.2021 05:10


Mathematics, 07.04.2021 05:10


Mathematics, 07.04.2021 05:10



Mathematics, 07.04.2021 05:10

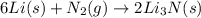
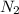 needed to react with 0.536 moles of Li.
needed to react with 0.536 moles of Li.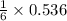 moles of
moles of ![N_2[tex] gas needed:[tex]=28 g/mol\times 0.0893 mol=2.5004 g](/tpl/images/0194/9373/6ce3e.png)
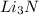
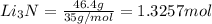
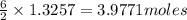
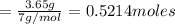
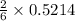 that is 0.1738 moles of
that is 0.1738 moles of 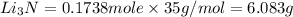
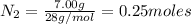
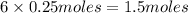 of lithium
of lithium

